All of our recommendations, unless specified, relate to acute COVID-19 in adults.
Some of our recommendations vary according to the severity of COVID-19 illness. Definitions of the categories are based on World Health Organization (WHO) criteria and can be viewed by clicking the plus (+) signs below.
RECOMMENDATION: The guidelines group does not recommend tocilizumab for non-severe or routinely for all people with severe or critical COVID-19. We suggest a single dose of tocilizumab (8mg/kg, maximum 800mg) to be added to corticosteroids in the absence of co-existing overt bacterial / fungal infection for people hospitalized with severe or critical COVID-19, within 24-48 hours of rapidly progressive respiratory failure, in the presence of significant systemic inflammation.
Note: significant systemic inflammation is usually characterized by clinical features such as high fever and elevated inflammatory markers, such as C-reactive protein of greater than 100mg/L (10mg/dL).
DATE OF RECOMMENDATION: 24th May 2021
Definition of non-severe:
NO features of severe or critical illness (see below). For some interventions this group may be divided into:
Mild illness
- Symptomatic (any acute COVID-19 related symptoms)
- AND respiratory rate <24/min
- WITHOUT pneumonia or hypoxia
Moderate illness
- Pneumonia (clinical or radiological) OR hypoxia (SpO2 <94% in adults with no underlying lung disease)
- AND respiratory rate ≤30/min
- AND SpO2 ≥90% on room air
A conditional recommendation is one for which the panel is less confident about the balance between desirable and undesirable effects of the intervention.
Definition of severe:
Pneumonia with ANY ONE of the following:
- respiratory rate >30/min
- severe respiratory distress
- SpO2 <90% on room air
- NO invasive or non-invasive respiratory support needed
Definition of critical:
- Requirement for high-level respiratory support: noninvasive ventilation, high-flow oxygen (≥20 litres per minute) or invasive mechanical ventilation
- OR acute respiratory distress syndrome (PaO2/FiO2 ratio of <300)
- OR sepsis
- OR shock
The evidence from 9 RCTs indicates that tocilizumab probably reduced mortality by a small magnitude. It also reduced progression to ventilation, ECMO or death in patients with severe or early critical COVID-19 with high levels of systemic inflammation and severe hypoxia. In the studies examined, most participants (>80%) were receiving steroids, had an average C-reactive protein of greater than 100 mg/L (10mg/dL), and needed oxygen therapy. One of the larger trials mandated initiation within 24 hours of intensive care admission. In these patients it may increase clinical improvement at 28 days and may reduce time to clinical improvement. The effects are modest.
The evidence was very uncertain on adverse events and serious adverse events, but the effect estimates did not suggest an increase in the arms receiving tocilizumab. Tocilizumab has a well-defined safety profile and is widely used for other conditions.
There is a lack of evidence for the use of tocilizumab in patients with non-severe COVID-19.
Therefore the panel judged that the evidence for modest benefit outweighs risk of harm in a narrowly-defined group of patients, hospitalized with severe or critical COVID-19, with evidence of significant systemic inflammation, and respiratory failure worsening despite use of systemic corticosteroid therapy.
Date of latest search: 21st April 2021.
Date of completion & presentation to Expert Working Group: 28th April 2021.
Date of planned review: 21st January 2022.
Evidence synthesis team: Anju Susan Jacob, Amita Jacob, Richard Kirubakaran, Bhagteshwar Singh and Priscilla Rupali.
Click here for an Interactive Summary of Findings table (opens in a new window).
GRADE Working Group grades of evidence
High certainty: We are very confident that the true effect lies close to that of the estimate of the effect
Moderate certainty: We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different
Low certainty: Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect
Very low certainty: We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect
Explanations:
a. Gordon REMAP-CAP 2021 [3]; Hermine CORIMUNO-19 2020 [4]; Horby RECOVERY 2021 [5]; Rosas COVACTA 2021 [6]; Salama EMPACTA 2020 [7]; Salavarani 2020 [8]; Soin COVINTOC 2021 [9]; Stone 2020 [10]; Veiga TOCIBRAS 2021 [11]
b. Not downgraded for risk of bias: We feel the imbalance in the proportions on Remdesivir, used as a co-intervention, in the two groups is unlikely to affect the outcome.
c. Not downgraded for indirectness: The majority of participants had severe or critical illness in the REMAP-CAP and RECOVERY trials, which contributed the most data to this meta-analysis.
d. Downgraded by 1 level for serious imprecision, since the lower bound (2% reduction) of the 95% CI includes clinical benefit that may not be appreciable in clinical practice.
e. Gordon REMAP-CAP 2021 [3]; Hermine CORIMUNO-19 2020 [4]; Horby RECOVERY 2021 [5]; Rosas COVACTA 2021 [6]; Salama EMPACTA 2020 [7]; Salvarani 2020 [8]; Stone 2020 [10]; Veiga TOCIBRAS 2021 [11]
f. Downgraded by 1 level for serious risk of bias: some concerns due to deviation from intended interventions, outcome measurement and selection of reported results
g. Downgraded by 1 level for serious indirectness, since the outcome is a composite outcome of in-hospital survival, hospital discharge, 2-point ordinal scale improvement
h. Gordon REMAP-CAP 2021 [3]; Hermine CORIMUNO-19 2020 [4]; Horby RECOVERY 2021 [5]; Rosas COVACTA 2021 [6]; Salama EMPACTA 2020 [7]; Salvarani 2020 [8]; Stone 2020 [10]
i. Gordon REMAP-CAP 2021 [3]; Hermine CORIMUNO-19 2020 [4]; Horby RECOVERY 2021 [5]; Salama EMPACTA 2020 [7]; Stone 2020 [10]; Veiga TOCIBRAS 2021 [11]
j. Hermine CORIMUNO-19 2020 [4]; Rosas COVACTA 2021 [6]; Salama EMPACTA 2020 [7]; Salvarani 2020 [8]; Soin COVINTOC 2021 [9]; Stone 2020 [10]; Veiga TOCIBRAS 2021 [11]; Wang 2021 [12]
k. Downgraded by 1 level for serious risk of bias: some concerns regarding randomization, deviations from intended interventions and outcome measurement
l. Downgraded by 1 level for serious inconsistency: I²=64%
m. Downgraded by 1 level for serious imprecision due to a wide confidence interval consistent with the possibility for no effect and the possibility for harm
n. Gordon REMAP-CAP 2021 [3]; Hermine CORIMUNO-19 2020 [4]; Rosas COVACTA 2021 [6]; Salama EMPACTA 2020 [7]; Salvarani 2020 [8]; Soin COVINTOC 2021 [9]; Stone 2020 [10]; Veiga TOCIBRAS 2021 [11]; Wang 2020 [12]
o. Downgraded by 1 level for serious risk of bias: some concerns regarding randomization, deviations from intended interventions and outcome measurement
p. Downgraded by 1 level for serious indirectness: the absolute estimates of serious adverse events varied between trials, for e.g. 30% to 40% for the Rosas COVACTA 2021 trial [6] and about 3% for the Gordon REMAP-CAP 2021 trial [3]
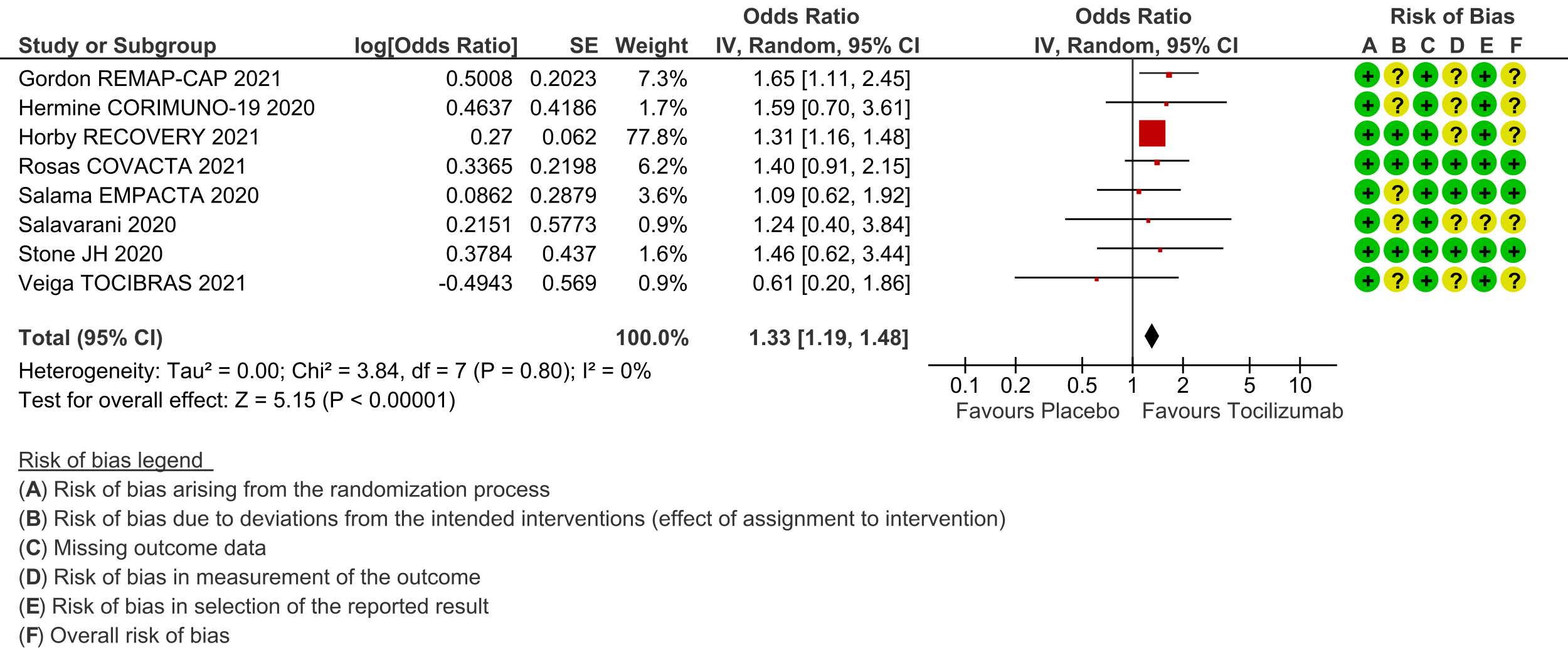
† For Clinical Improvement, as the event rate is >20% [common event], OR may overestimate. Hence we calculated RR with 95% CI – 1.14 [1.08, 1.18].
The incidence of COVID-19 infection in India has reached unprecedented proportions leading to considerable mortality and morbidity among those infected. Health care systems are facing enormous pressures with regard to oxygen supplies, admission of patients into intensive care, and what are considered lifesaving drugs. Death due to COVID-19 is believed to be related to immune dysregulation, often leading to acute inflammation, a cytokine storm leading on to an acute lung injury, acute respiratory distress syndrome (ARDS), coagulopathy and multi-organ dysfunction.
Markedly elevated inflammatory markers (such as C-reactive protein, D-dimer and ferritin) and pro-inflammatory cytokines like IL-6 are associated with critical and fatal COVID-19 and it is believed that blocking this inflammatory pathway may prevent disease progression [1]. There is therefore, a huge demand for drugs that will improve outcomes even marginally.
A scoping search revealed many published systematic reviews investigating use of Tocilizumab in COVID-19. It was decided to select a high-quality systematic review to inform this evidence review. The criteria for selection of this systematic review was based on the following criteria:
- Included and only meta-analysed data from randomized controlled trials
- Had a recent latest search date (within the last three months)
- Included all available outcomes
- Matched the pre-defined PICO question closely
- Scored a high or moderate score on the AMSTAR 2 quality assessment tool
To identify all available systematic reviews pertaining to our PICO question, a systematic search of Pubmed, Cochrane and the Epistemonikos databases was conducted. The search strategy was designed and validated by the Group’s information specialist. The search results were screened independently by two reviewers against the pre-defined criteria set out above, using Rayyan. The reviews identified were prioritised based on quality according to the AMSTAR 2 score and the most recent search date.
A systematic search was also done to identify studies meeting the inclusion criteria for the review but published after its latest search. As this review will inform a recommendation for India, a separate search was conducted for studies of any design investigating use of Tocilizumab for COVID-19 in India.
Where possible, we used data extracted by authors of the selected review. Where outcomes data were not found in the review at all or in the format required, we re-extracted them from the primary trial reports directly, for each of the outcomes identified as critical or important by the Expert Working Group prior to commencing the review:
Critical (Primary) outcomes:
- All-cause mortality
- Clinical improvement at day 28
- Time to clinical recovery
- Progression mechanical ventilation/ECMO/death
- Any adverse events
- Serious adverse effects
Important (Secondary) outcomes
- Admission to critical care
- Time to ventilation
- Duration of ventilation
- Length of hospital stay
- Length of stay in critical care
- Organ support other than ventilation
- Change in CRP at 48 hours (as a percentage of the initial value)
We did not conduct a separate risk of bias assessment as it had already been performed in duplicate as part of the systematic review selected.
We performed our own meta-analysis using Review Manager (RevMan) 5.4 using a random‐effects model for each of the critical and important outcomes where pooling of effect estimates was appropriate. We used risk ratios (RR) for dichotomous outcomes and mean differences (MD) for continuous outcomes, with 95% confidence intervals (CIs). We used the I2 statistic to measure residual heterogeneity. We used GRADE methodology to assess the certainty in the evidence, and documented this in a ‘Summary of findings’ table using GradeProGDT.
The review selected was the collaborative living network meta-analysis at covid-nma.org, which is published in the Cochrane Database of Systematic Reviews periodically as a review of IL-6 inhibitors for COVID-19, therefore including both tocilizumab and sarilumab [2].
Our updated search found 10 trials [3-12], that were already included in the COVID-NMA/Cochrane review, including an Indian study by Soin et al [12]. All participants were hospitalised adults, with an average age ranging from 54 to 65 years. There one trial reporting from each of Brazil, China, France, India, Italy, UK and USA; three were multi-country trials. Disease severity was mostly severe or critical (though some participants in the RECOVERY 2021 trial whom we labelled as at least moderate by the guidelines group’s criteria would probably be classified as severe [5]). The median or mean C-reactive protein was around or above 100mg/L in 8/10 trials, indicating significant systemic inflammation. Prevalence of comorbidities varied substantially between trials. Most trials used weight-based dosing of tocilizumab, up to a maximum of 800mg per dose; seven of the 10 trials offered treating clinicians the option of a second dose, though in trials that reported the numbers receiving this, a minority of participants received two doses. Most (7/10) trials were unblinded. Use of co‐interventions varied substantially between trials. The ‘Summary of characteristics of included trials’ table summarises key information about the trials; further detailed characteristics are available in the Cochrane review [2].
Most trials were judged to be at low risk of bias or had some concerns about a few domains; none were at high risk of bias overall for the outcomes considered. Risk of bias for each domain per trial is displayed alongside the Forest plots below.
The effect of Tocilizumab was evaluated for critical outcomes as follows.
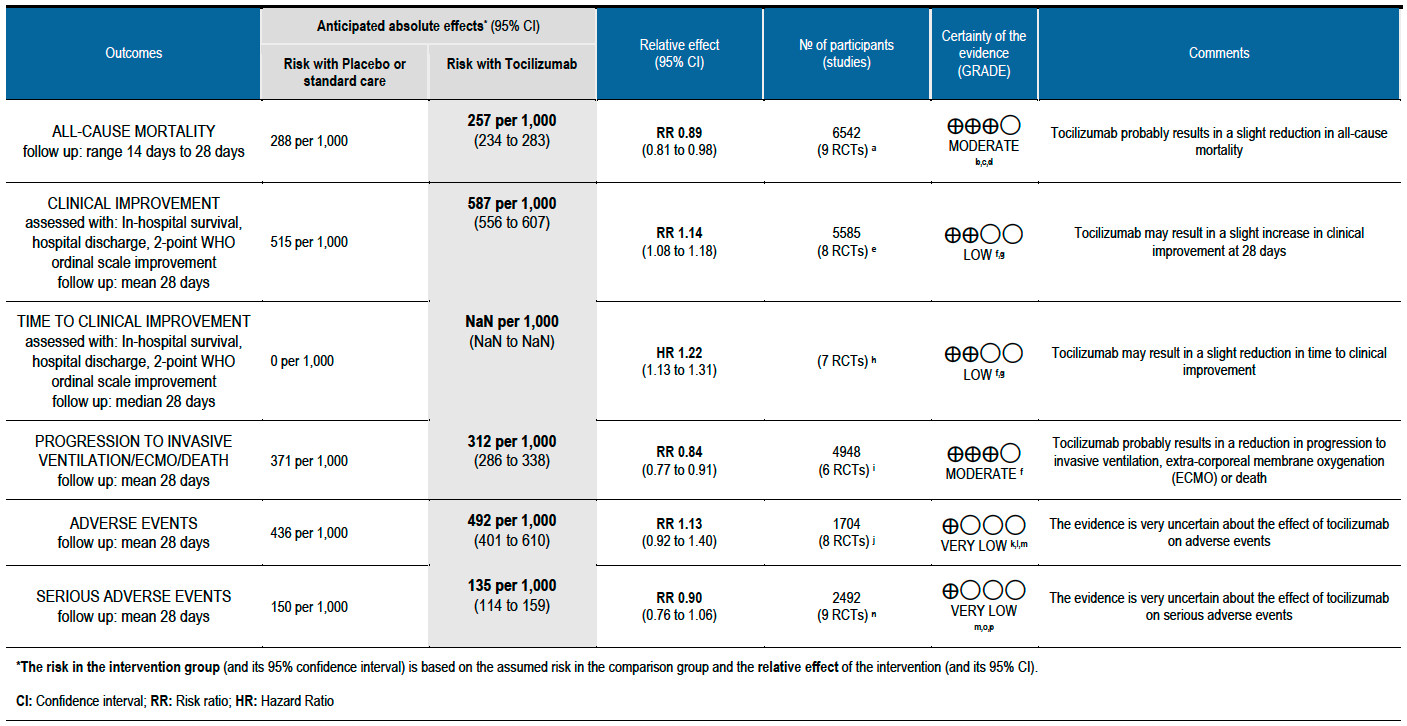
All-cause mortality (follow up till 14-28 days): Moderate certainty evidence from 9 RCTs [3-11] that included 6542 people with severe to critical categories of COVID-19 disease indicates that tocilizumab added to corticosteroids reduced all-cause mortality on average by 11% (95% CI 19% to 2%) compared to usual care and steroids. Based on the data, we anticipate that for 1000 people with severe and critical COVID disease treated with placebo or standard of care (that includes steroids) 288 deaths due to any cause are likely. We anticipate that there would probably be 32 fewer (from 6 fewer to 55 fewer) deaths per 1000 people if tocilizumab is added to steroids and standard care in people with severe to critical COVID disease.
Clinical improvement (at day 28; defined as in-hospital survival, hospital discharge or 2-point improvement on the WHO ordinal scale): Low certainty evidence from 8 RCTs [3-8;10;11] with 5585 severe and critically ill adult in-patients with COVID-19 suggests that tocilizumab added to steroids may increase the odds of clinical improvement compared to placebo or usual care with steroids (odds ratio [OR] 1.33; 95% CI 1.19 to 1.48). For every 1000 patients treated with placebo or usual care and steroids, 515 are likely to clinically impove. Adding tocilizumab may result in 72 more (from 41 more to 93 more) per 1000 people showing clinical improvement. As this event was common (>20%), in which case OR may overestimate an effect size, we calculated RR, which was 1.14 (95% CI 1.08 to 1.18).
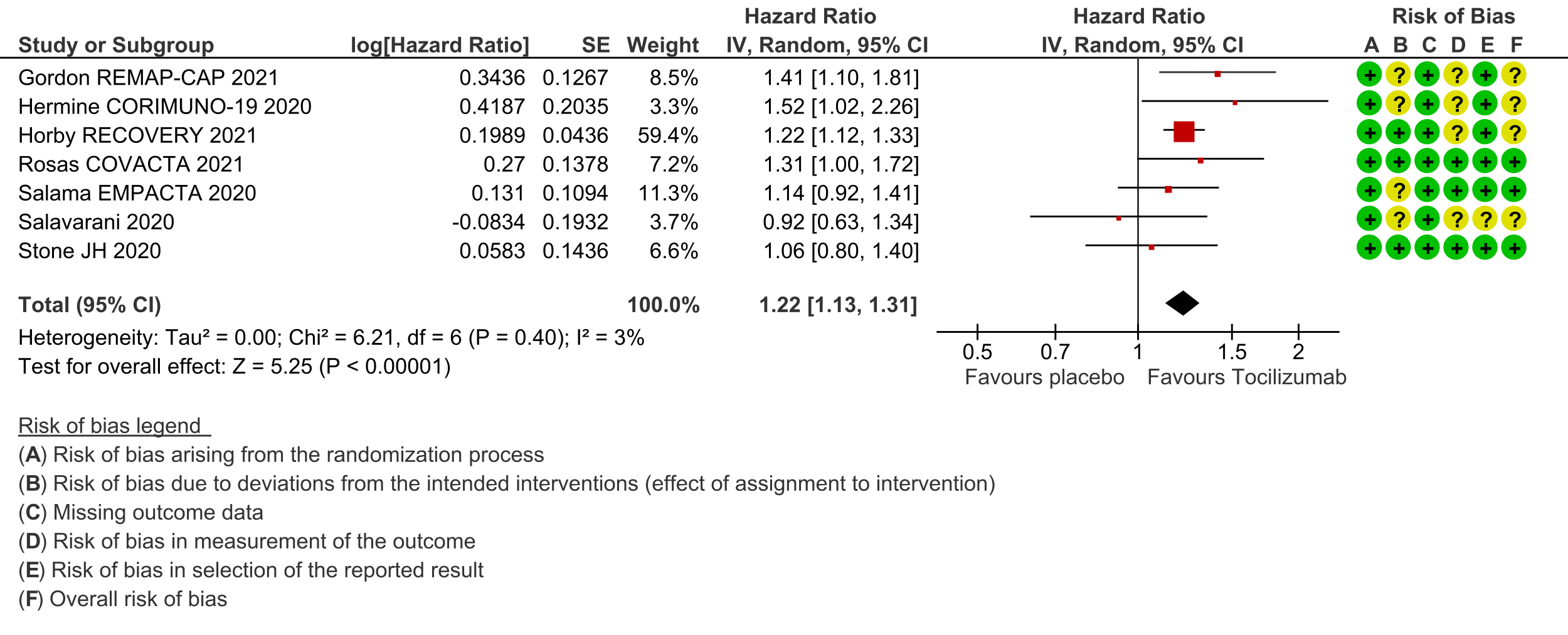
Time to clinical improvement (follow up median 28 days (defined as in hospital survival, hospital discharge or 2-point improvement on the WHO ordinal scale): Low certainty evidence from 7 RCTs [3-8;10] suggests that the time to clinical improvement was reduced by around one day when tocilizumab was added to steroids (medians ranged from 6 to 20 days) compared to standard care and steroids (5 to >28 days) [hazard ratio (HR) 1.22; 95% CI 1.13 to 1.31].
Progression to invasive ventilation or extra-corporeal membrane oxygenation (ECMO) or death (these were reported as a composite outcome in one large trial [3]): Moderate certainty evidence from 6 RCTs [3-5;7;10;11] indicates that from among 4948 severe or critically ill patients with COVID-19, the proportions progressing to invasive ventilation, extra-corporeal membrane oxygenation (ECMO) or death (due to not being mechanically ventilated) was reduced on average by 16% (95% CI 23% to 9%) when tocilizumab was added to steroids and usual care compared to only usual care and steroids. This suggests that for every 1000 patients who are severe or critically ill with COVID-19 and treated with standard care and steroids but not given tocilizumab, 371 will probably progress to this outcome compared to 59 fewer (from 85 fewer to 33 fewer) per 1000 patients also given tocilizumab.
Adverse events (follow up till day 28): Two trials (REMAP-CAP [3] and RECOVERY [5]) did not report adverse events. Very low certainty evidence from 8 RCTs [4; 6-12] that included 1704 severely or critically ill patients suggested that adverse events may be little or no different when tocilizumab was added, or not added, to steroids and usual care (RR 1.13; 95% CI 0.92 to 1.40).
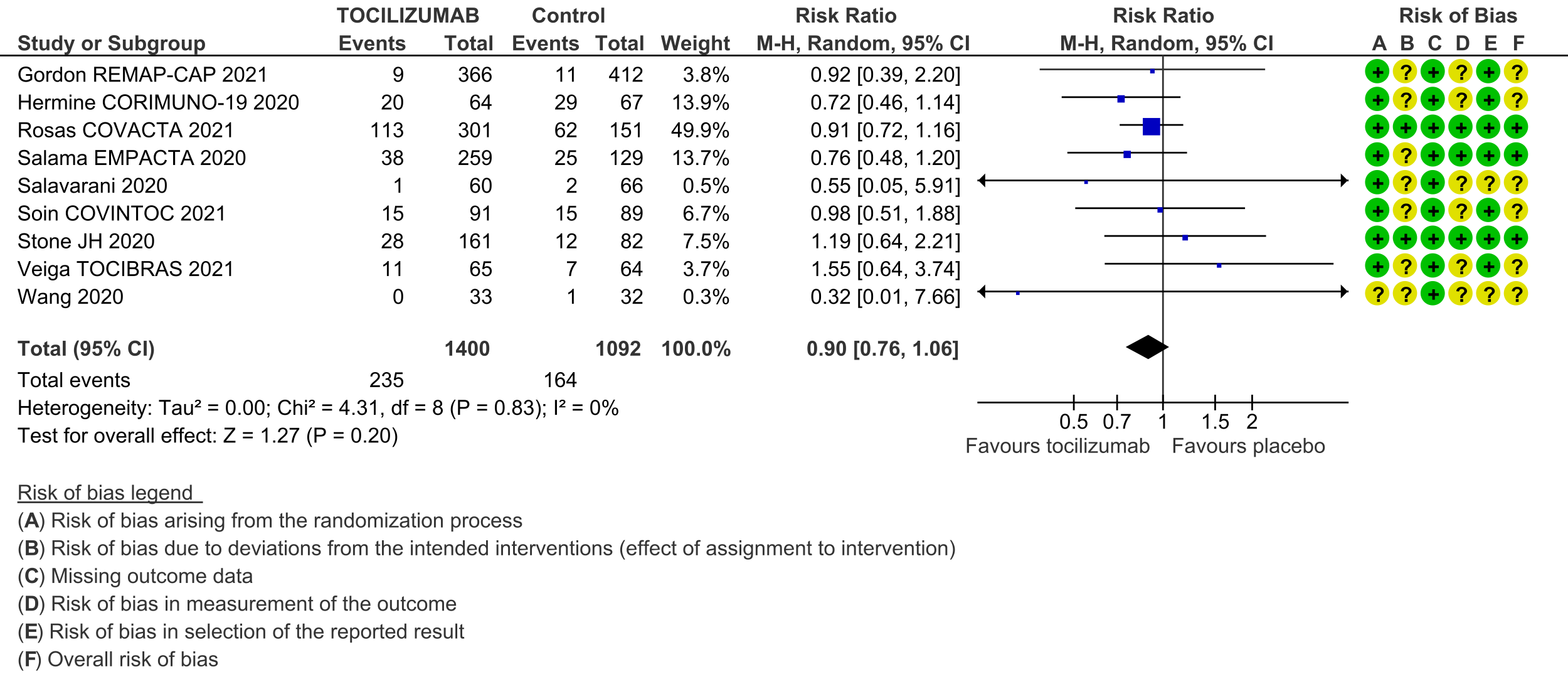
Serious adverse events (follow up till day 28): These were reported by 9 RCTs [3;4;6-12]. There was again very low certainty in the evidence, though the effect estimate from meta-analysis found no significant difference between the tocilizumab and control groups: RR 0.9 (95% CI 0.76 to 1.06.
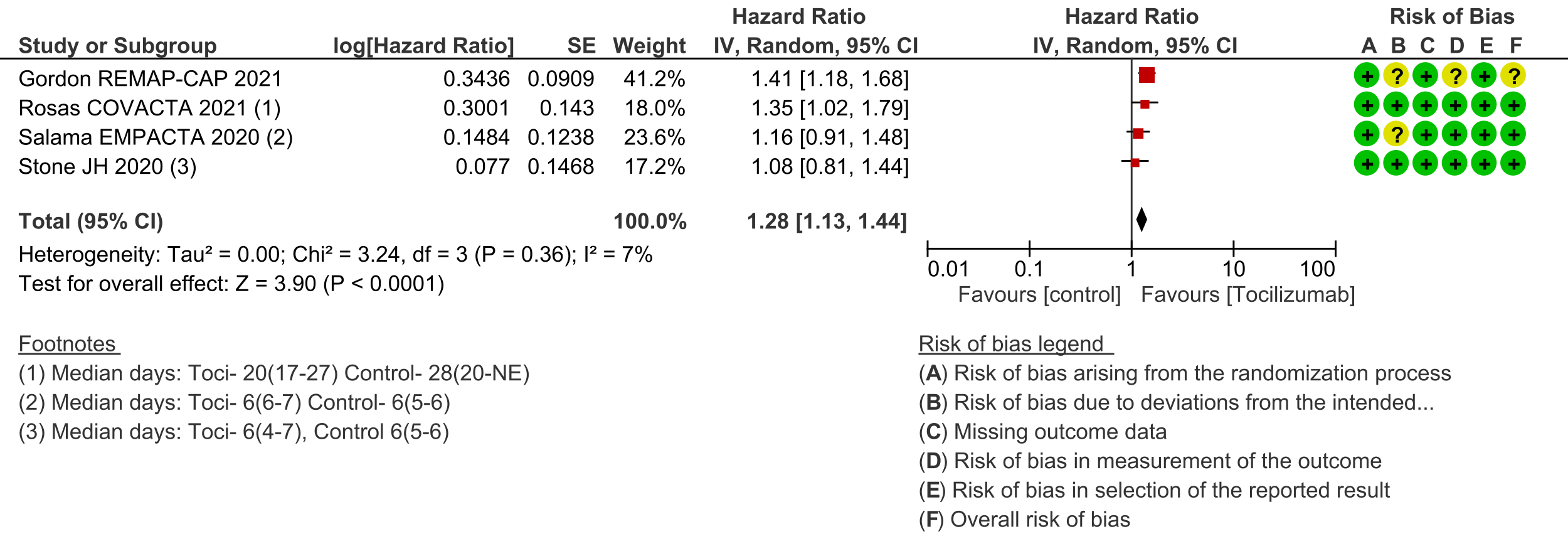
Secondary outcomes: Length of hospital stay favoured the tocilizumab group with a HR of 1.28 (4 trials [3;6;7;10]; 95% CI 1.13 to 1.44). Admission to critical care was not different between the two groups (OR 0.8; 2 trials [6;9]; 95% CI 0.50 to 1.28). Ventilator-free days and length of stay in critical care were variably expressed as median, rate ratio or adjusted odds ratio and hence could not be pooled. However, the median difference of ventilator-free days varied from 0 to 5.5 days shorter in the tocilizumab groups in 4 trials [3;6;9;11], and length of critical care stay in 3 trials ranged from a median difference of 2 to 5.5 days shorter in the tocilizumab groups [3;6;9]. Secondary infections in participants in the tocilizumab groups ranged from 1.7% to 20.6%; in the control groups this was 5.7% to 24.5%; none of the trials reported a higher incidence of secondary infections in the tocilizumab group [4;6-11].
Subgroup analyses: While formal subgroup analyses could not be performed, we attempted to inform the optimal use of tocilizumab using the following approaches:
(a) We found that the median CRP across the studies varied, ranging widely from 6-160mg/L. The median CRP was >100 mg/L for the two largest trials: it was 150mg/L in REMAP-CAP [3] and 143mg/L in RECOVERY [5]. The other inflammatory marker evaluated was Ferritin in 2 trials [8;11], which was not reported to demonstrate any meaningful correlation.
(b) REMAP-CAP was the only study to document time to administration of tocilizumab after admission to a critical care unit, with a median of 13.6 hours [3]. Time of administration since symptom onset was also noted in 5 trials and varied from a median of 8 to 11 days [3;4;6;8;10].
(c) To determine efficacy with steroids vs. no steroids: this outcome was reported in subgroup analyses performed by 2 trials [4;7]. Both favoured administration of Tocilizumab with steroids though these trials had fewer numbers with a low event rate. In addition, >80% of participants in the 2 largest trials had Tocilizumab added on to their steroids [3;5].
(d) Data were not available for other subgroup analyses for time since onset of respiratory support (though see point (a) above), lung involvement on CT scan, single vs. two doses, and other markers like procalcitonin and pentraxin.
| Study |
Intervention & comparator arms |
Design |
Location |
Age, average in years |
No. of participants randomized |
Participant characteristics |
|---|---|---|---|---|---|---|
| Gordon REMAP‐CAP 2021 [3] |
Tocilizumab (8 mg/kg, maximum 800 mg; (Note that the sarilumab |
Unblinded RCT |
Australia, Ireland, Netherlands, |
Tocilizumab: 61.5 Standard care: 61.1 |
Tocilizumab: 353 Standard care: 402 |
COVID-19 severity: 69% severe; 28% Median CRP: 130-150 mg/L |
| Hermine CORIMUNO‐19 2020 [4] |
Tocilizumab (8 mg/kg; optional 2nd |
Unblinded RCT |
France |
64.8 |
Tocilizumab: 64 Standard care: 67 |
COVID-19 severity: 42% moderate; 58% Median CRP: 120-127 mg/L |
| Horby RECOVERY 2021 [5] |
Tocilizumab (400-800 mg depending on weight; optional 2nd dose after 12-24 hours - number |
Unblinded RCT |
UK |
63.6 |
Tocilizumab: 2022 Standard care: 2094 |
COVID-19 severity: least 41% severe; at least 13% critical Median CRP: 143-144 mg/L (> 75 was an |
| Rosas COVACTA 2021 [6] |
Tocilizumab (8 mg/kg, maximum 800 mg; |
Double-blinded RCT |
Canada, Denmark, France, Germany, Italy, |
60.8 |
Tocilizumab: 301 Standard care: 151 |
COVID-19 severity: least 14% severe; 25% critical Median CRP: 150-157 mg/L |
| Salama EMPACTA 2020 [7] |
Tocilizumab (1 dose of 8 mg/kg, maximum |
Double-blinded RCT |
Brazil, Kenya, Mexico, Peru, South Africa, |
55.9 |
Tocilizumab: 259 Standard care: 129 |
COVID-19 severity: 62% moderate; 26% Median CRP: 125-143 mg/L |
| Salvarani 2020 [8] |
Tocilizumab (8 mg/kg, maximum 800 mg; optional |
Unblinded RCT |
Italy |
60 |
Tocilizumab: 60 Standard care: 66 |
COVID-19 severity: all severe Median CRP: 6.5-10.5 |
| Soin COVINTOC 2021 [9] |
Tocilizumab (6 mg/kg, maximum 480 mg; |
Unblinded RCT |
India |
Tocilizumab: 56 Standard care: 54 |
Tocilizumab: 91 Standard care: 88 |
COVID-19 severity: 40% moderate; 50% Mean CRP: 88-110 mg/L |
| Stone 2020 [10] |
Tocilizumab (1 dose of 8 mg/kg, maximum |
Double-blinded RCT |
USA |
60 |
Tocilizumab: 161 Standard care: 82 |
COVID-19 severity: 80% moderate; 4% Median CRP: 94-116 mg/L (CRP > 50 mg/L |
| Veiga TOCIBRAS 2021 [11] |
Tocilizumab (1 dose of 8 mg/kg, maximum |
Unblinded RCT |
Brazil |
57.4 |
Tocilizumab: 65 Standard care: 64 |
COVID-19 severity: 52% moderate; 32% Mean CRP: 160-193 mg/L |
| Wang 2020 [12] |
Tocilizumab (400 mg; optional 2nd dose after |
Unblinded RCT |
China |
63 |
Tocilizumab: 33 Standard care: 32 |
COVID-19 severity: 57% moderate; 43% Median CRP: 6-10 mg/L |
1. Mortality, all-cause: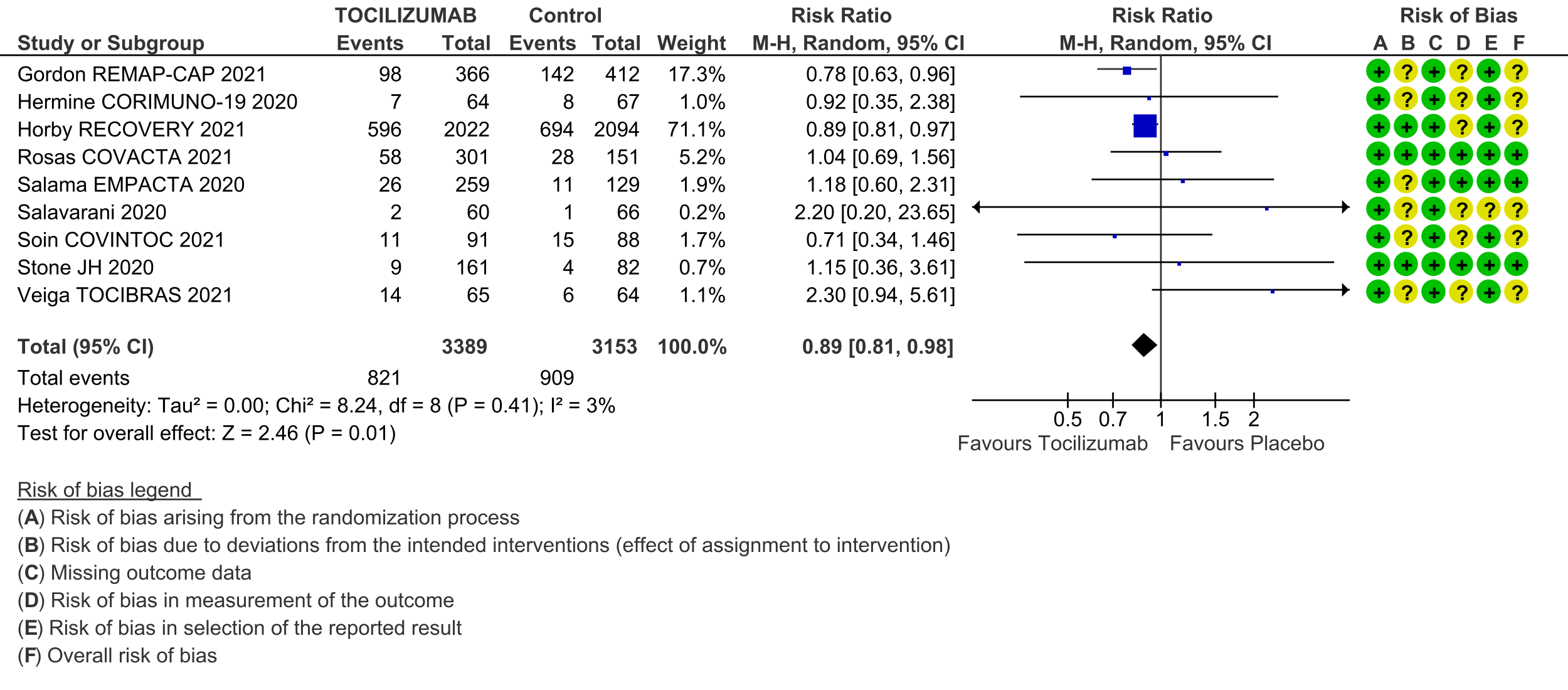
2. Clinical improvement:
3. Time to clinical improvement:
4. Progression to invasive ventilation, ECMO or death:
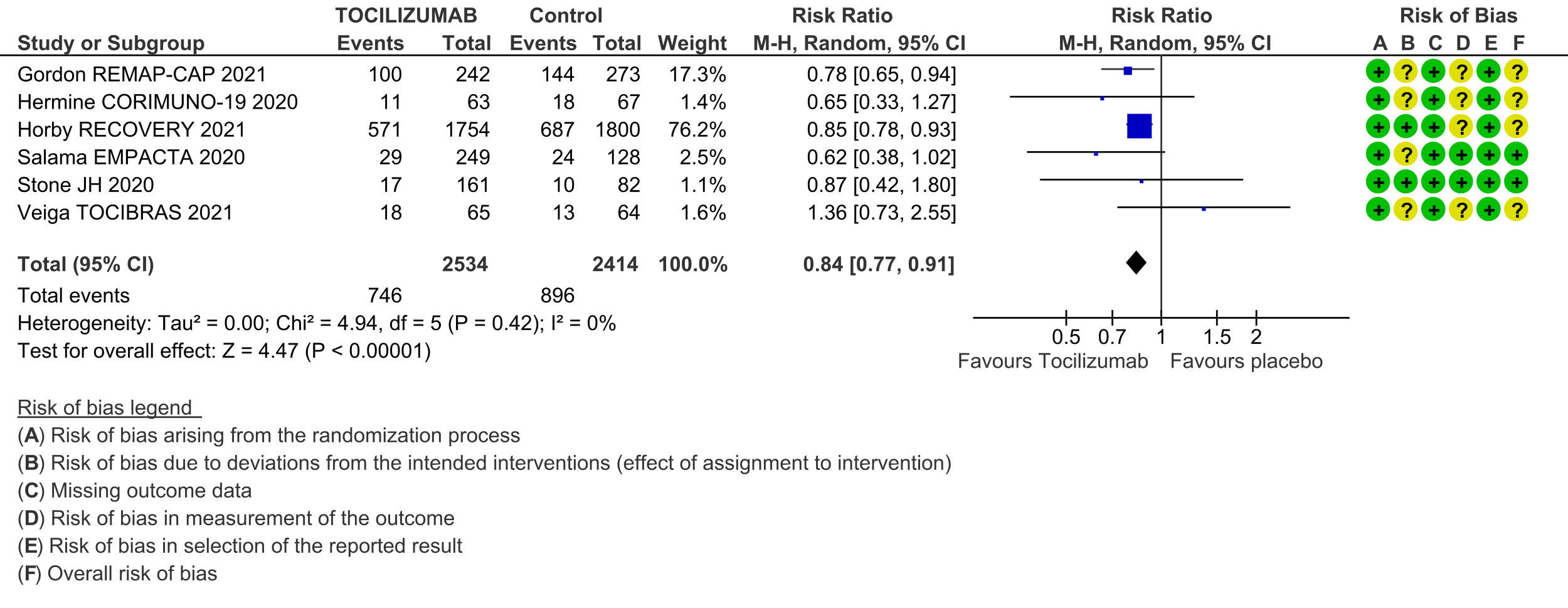
5. Adverse events:
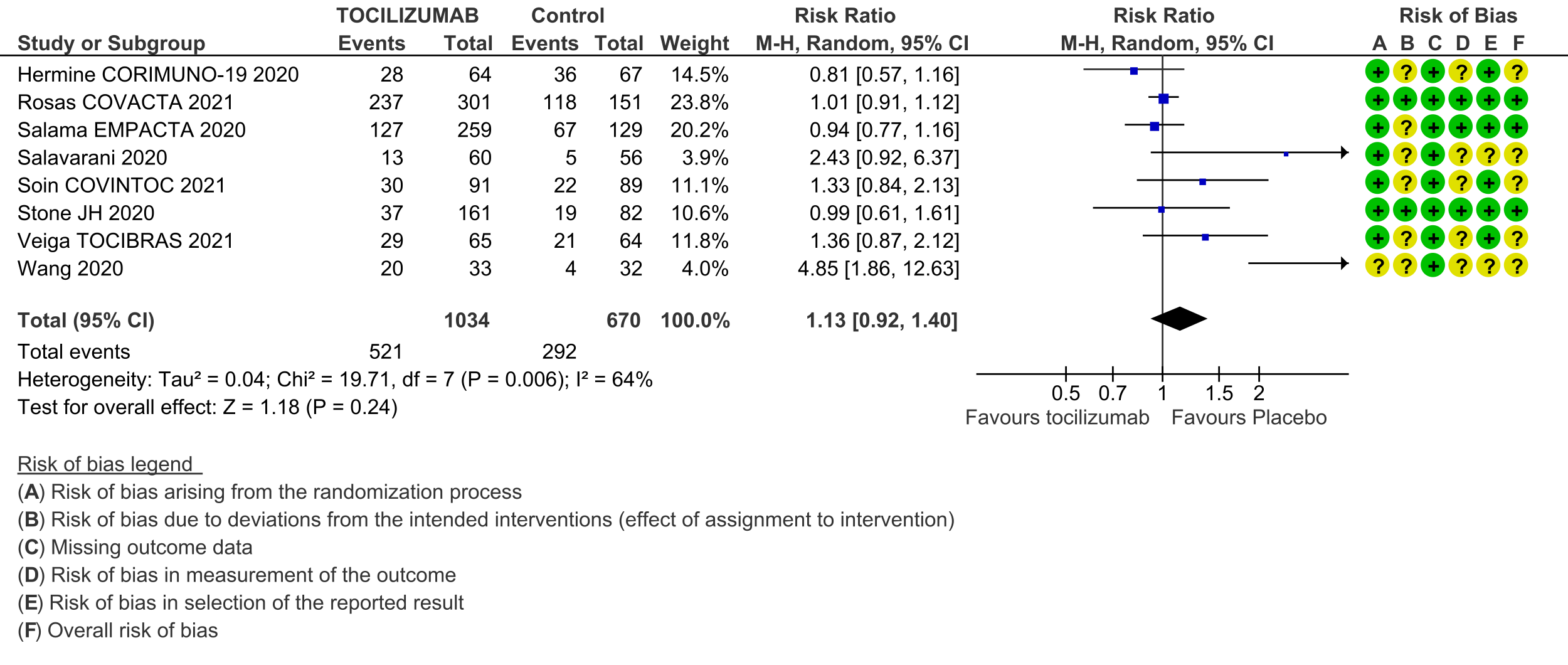
6. Serious adverse events:
7. Length of stay in hospital:
8. Admission to critical care:
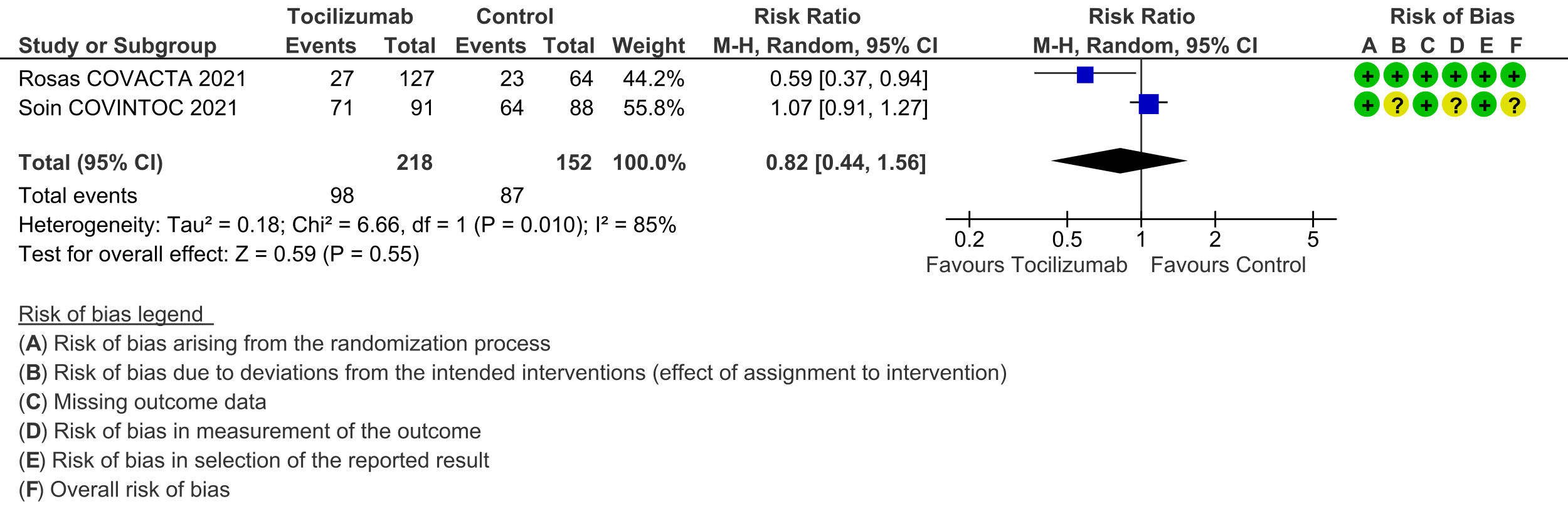
The Anti-inflammatory Expert Working Group met on 28th April 2021 to consider tocilizumab as a treatment for COVID-19. Conflict of interest declarations were reviewed by the Steering Committee; none were found to be relevant to tocilizumab.
A summary and then more detailed explanations of the Expert Working Group's judgements follow.
Summary of judgements
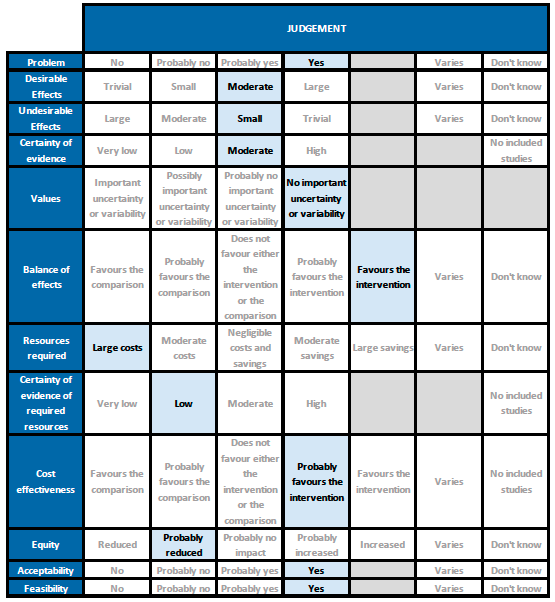
Problem
COVID-19 infection continues to wreak havoc in India and it is likely that the second surge will cause significant mortality and morbidity. Hence therapeutic interventions for clinical management are a priority. Today with the oxygen needs climbing everywhere in the country we need resources and drugs which will reduce the duration of severe/critical disease. However, the subset of patients who will benefit with tocilizumab needs to be very carefully defined, in view of the direct and indirect costs associated with this drug.
Desirable effects
Tocilizumab reduces mortality on average by 11% (95% CI 2% to 19%), increases clinical improvement by 14% (95% CI 8% to 18%), reduces progression to invasive ventilation by 16% (95% CI 9% to 23%), and reduces the time to clinical improvement and to hospital discharge. The expert working group (EWG) noted that these effects were of moderate significance. The EWG felt that reducing progression of ventilation and improving time to clinical recovery were important outcomes, which would help prevent admissions and intensive care costs and save on oxygen, which is currently in acute short supply.
Undesirable effects
The pooled data did not suggest any increase in serious or other adverse events when tocilizumab was added to usual care that included corticosteroids. Secondary infections were also uncommon, though these were not reported by all trials, and follow-up in most trials was until 14 or 28 days, which might be early for development of some secondary infections. It should be noted that the impact of a secondary infection may be greater in India, where nosocomial infections are often due to multi-drug resistant organisms, which might be more difficult to treat. Concurrent active infection with another pathogen was an exclusion criterion for most trials. Hence the expert working group felt that the undesirable effects may be small.
Certainty of evidence
Using GRADE methodology the evidence synthesis team rated the certainty of evidence as moderate for all cause mortality and progression to ventilation/ECMO/death; low certainty for clinical improvement and time to clinical improvement; and very low for adverse events and serious adverse events. The expert working agreed with these judgements and rated the overall certainty of evidence as moderate.
Values
The outcomes of death, clinical improvement at day 28, time to clinical improvement, progression to mechanical ventilation were variably expressed by different studies. However, the expert working group felt that there was no significant variability or uncertainty in how people would value the main outcomes. The expert working group felt that reducing progression of ventilation and improving time to clinical recovery would help prevent admissions and intensive care costs and save on oxygen consumption which is in severely short supply.
Balance of effects
The expert working group felt that the balance of effects favoured the intervention. Given that the country is facing a shortage of beds, oxygen and intensive care units, if an intervention could reduce death, progression to mechanical ventilation and time to clinical improvement it would be of enormous benefit. The expert working group also recognized that this intervention is expensive and applicability of this intervention in a secondary care setting is more difficult.
Resources required
The expert working group recognized that there are large direct and indirect costs and resources required to deliver Tocilizumab as an intravenous injection.
Certainty of evidence of required resources
The expert working group acknowledged that there may be no studies evaluating required resources and hence felt no judgment on certainty could be made. However they did acknowledge there maybe cost savings on implementation of this intervention vis-a-vis intensive care beds, the need for ventilation, and other resource benefits.
Cost effectiveness
Published evidence was not consulted for this judgement, but the expert working group also acknowledged that this intervention might be variably used in private not-for-profit, private for-profit and public hospitals due to the attendant costs. However, the working group felt the cost-effectiveness probably favoured the use of tocilizumab in a carefully selected sub group of patients with serious or critical COVID-19 disease.
Equity
The working group felt that impact of the use of tocilizumab would probably reduce equity. This is an expensive intervention and hence it is likely to create inequity between those who can afford it versus those who cannot, and in places where it is available freely versus where it is not.
Acceptability
The expert working group felt that, apart from the cost, this is an acceptable intervention as it is an intravenous single dose injection that could reduce mortality without an increase in adverse events.
Feasibility
The working group felt that is feasible to implement this intervention, as tocilizumab has been used widely before in the treatment of chronic rheumatic diseases.
Tocilizumab should only be added on to corticosteroid therapy. The largest trials reported a median CRP >100mg/L (>10mg/dL) and oxygen need. Treatment with tocilizumab was initiated within 24 hours after admission to intensive care in one of the largest trials. Quicker clinical improvement and reduced progression to ventilation with Tocilizumab in these patients may lead to increased hospital bed availability and reduced oxygen use. However, it is an expensive intervention and in short supply, so physicians who prescribe this intervention need to consider the additional benefits over corticosteroids vs. the cost burden it entails.
Tocilizumab should NOT be used in a patient who has an active clinically evident bacterial, mycobacterial, viral or fungal infection as previous experience with use in autoimmune conditions suggests that it may contribute to an increased risk of secondary infections [13]. However in the meta-analysis of RCTs studied we did not encounter an increased risk of secondary infections. There are numerous reports of invasive fungal infections and tuberculosis in patients with COVID-19, which are more common when corticosteroids are used, and their risk might be increased further by tocilizumab use. This may be of greater concern for patients with diabetes mellitus (especially with poor glycaemic control), who are immunosuppressed (including due to high-dose corticosteroids) and those who are neutropenic. Thus we recommend taking into consideration very carefully the type of host and the entire clinical setting to encourage responsible use, and these groups may need closer monitoring.
After administration, it may be prudent to evaluate for infusion reactions, hepatitis, thrombocytopenia, neutropenia and hyperglycaemia and onset of secondary infections.
The expert working group recommends tocilizumab in a specific subgroup of patients with rapidly progressive clinical worsening of severe or critical COVID-19 with evidence of significant systemic inflammation and high oxygen requirement. Disaggregated data for the effects of tocilizumab were not available from the trials that did include individuals from the pre-specified subgroups, particularly children, pregnant women and those with chronic kidney or liver disease.
Based on product literature and use for other conditions, tocilizumab should be used with more careful monitoring in patients at high risk of gastrointestinal perforation, and those with hepatic impairment, demyelinating disorders, thrombocytopenia or neutropenia. Pregnancy data from animals shows an increased rate of congenital malformations and possibly pre-term birth or spontaneous abortions. However, it has been used successfully in pregnancy during this pandemic and so a risk/benefit discussion should take place between clinician and patient. Tocilizumab has also been used widely in children for other conditions, so a risk/benefit discussion should take place regarding its use for children.
Use of tocilizumab and real-world safety should be monitored using audits and registries. In particular, its use in the subgroups mentioned above, and incidence of adverse effects, should be prioritised.
- Though it has been shown that Tocilizumab may improve clinical outcomes when added on to corticosteroids, applicability will only be evident after a cost-benefit analysis in resource limited settings in India. This is an urgent research priority considering the country is facing a manpower, oxygen and intensive care unit bed shortage.
- In addition, the utility of biomarkers such as CRP predicting response in an Indian setting should be studied.
- Further randomized controlled trials should be conducted to enable further precision on the optimal timing for use of Tocilizumab in the course of the COVID-19 illness, and the patient subgroup with maximum benefit and least harm.
- Mehta P, McAuley DF, Brown M, et al. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet 2020; 395:1033.
- Ghosn L, Chaimani A, Evrenoglou T, et al. Interleukin‐6 blocking agents for treating COVID‐19: a living systematic review. Cochrane Database of Systematic Reviews 2021, Issue 3. Art. No.: CD013881. DOI: 10.1002/14651858.CD013881.
- Investigators TR-C, Gordon AC, Mouncey PR, et al. Interleukin-6 Receptor Antagonists in Critically Ill Patients with Covid-19 – Preliminary report. medRxiv. 2021 Jan 7;2021.01.07.21249390.
- Hermine O, Mariette X, Tharaux P-L, et al. Effect of Tocilizumab vs Usual Care in Adults Hospitalized With COVID-19 and Moderate or Severe Pneumonia: A Randomized Clinical Trial. JAMA Intern Med. 2021 Jan 1;181(1):32–40.
- Group RC, Horby PW, Pessoa-Amorim G, et al. Tocilizumab in patients admitted to hospital with COVID-19 (RECOVERY): preliminary results of a randomised, controlled, open-label, platform trial. medRxiv. 2021 Feb 11;2021.02.11.21249258.
- Rosas IO, Bräu N, Waters M, et al. Tocilizumab in Hospitalized Patients with Severe Covid-19 Pneumonia. N Engl J Med. 2021 Apr 22;384(16):1503–16.
- Salama C, Han J, Yau L, et al. Tocilizumab in Patients Hospitalized with Covid-19 Pneumonia. N Engl J Med. 2021 Jan 7;384(1):20–30.
- Salvarani C, Dolci G, Massari M, et al. Effect of Tocilizumab vs Standard Care on Clinical Worsening in Patients Hospitalized With COVID-19 Pneumonia: A Randomized Clinical Trial. JAMA Intern Med. 2021 Jan 1;181(1):24.
- Stone JH, Frigault MJ, Serling-Boyd NJ, et al. Efficacy of Tocilizumab in Patients Hospitalized with Covid-19. N Engl J Med. 2020 Dec 10;383(24):2333–44.
- Veiga VC, Prats JAGG, Farias DLC, et al. Effect of tocilizumab on clinical outcomes at 15 days in patients with severe or critical coronavirus disease 2019: randomised controlled trial. BMJ. 2021 Jan 20;372:n84.
- Wang D, Fu B, Peng Z, et al. Tocilizumab in patients with moderate or severe COVID-19: a randomized, controlled, open-label, multicenter trial. Front Med. 2021. https://doi.org/10.1007/s11684-020-0824-3
- Soin AS, Kumar K, Choudhary NS, et al. Tocilizumab plus standard care versus standard care in patients in India with moderate to severe COVID-19-associated cytokine release syndrome (COVINTOC): an open-label, multicentre, randomised, controlled, phase 3 trial. Lancet Respir Med. 2021 May 1;9(5):511–21.
- Morel J, Constantin A, Baron G et al. Risk factors of serious infections in patients with rheumatoid arthritis treated with Tocilizumab in the French registry (REGATE). Rheumatology 2017;56:17461754. doi:10.1093/rheumatology/kex238.
Covid Management Guidelines India Group - Anti-inflammatory Working Group. Tocilizumab. Covid Guidelines India; Published online on May 24, 2021; URL: https://indiacovidguidelines.org/tocilizumab/ (accessed ).
